Medical Research
Lexaria Releases 2018 Human Clinical Study Results on CBD Delivery and Effectiveness in Medical Journal

- The findings of the human clinical trial were published in Advances in Therapy.
- The company found that, with their TurboCBD™ 90 mg dose, the subjects experienced high CBD plasma levels that maintained their concentrations evenly after the 30-minute mark.
The cannabis world is largely struggling with legalization due to one big problem – research. Regardless of the new drug reform bills on the desks of senators, or the legalization of hemp through the 2018 Farm Bill, there is still so much that the public doesn’t actually know about hemp and cannabis. Lexaria Bioscience Corp., an innovator in the drug delivery sector, wants to face that problem head on, which they’ve done through a human clinical study in 2018, which examined CBD delivery and efficacy with their own DehydraTECHTM powered TurboCBD™ capsules.
Lexaria recently published a press release to announced that their final study results are available and have already been published in a respected medical journal called Advances in Therapy. This publication is known for their longevity in the world of clinical medicine and pharmaceutical research, as they’ve been continually publishing in this sector for over three decades.
In the publication, Lexaria describes the findings of their clinical trial. The key findings revealed:
- The 90 mg dose of TurboCBD™ caused higher CBD in-plasma levels than the generic doses at both 90-minute and 120-minute status checks.
- The only CBD dose that was elevated at 30 minutes and remained elevated was the TurboCBD™ 90 mg dose.
- The 90 mg TurboCBD™ dose coincided with a decrease in mean arterial blood pressure, compared to baseline.
- Relative to baseline, the TurboCBD™ 90 mg dose showed a significantly higher cerebral perfusion, which was “analyzed by an index of conductance in the middle cerebral artery (MCA),” according to the study.
The study specifically involved 12 healthy young male athletes, and the results were promising. However, to gain a better understanding of what these products can do, the investigators believe that additional research would be helpful. More specifically, they stated, “further acute and chronic dosing investigations in older and cerebrovascular-compromised patients may shed light on the vascular and clinical impacts of increased CBD bioavailability.”
Lexaria was recently the recipient of two patents that could greatly benefit their research in the future. The first was granted for “treating certain conditions including, but not limited to heart disease, neurological diseases such as Alzheimer’s, Parkinson’s, schizophrenia,” and other conditions. The decision to grant this patent is fairly convenient, due to how effective that the DehydraTECH technology is proving to be in the increased cerebral blood flow. The study’s success in delivering drugs to the brain is also a major milestone, which could help millions of people with these conditions in the future.
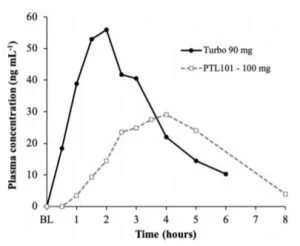
In the publication, another recent study was discussed by other investigators that examined CBD with the absorption that human experienced with edibles. The chart above shows the differences between the Turbo CBD 90 mg dose and that of the competitors 100 mg dose. While the latter took about four hours to reach maximum plasma CBD concentration, the TurboCBD™ 90 mg dose took a maximum of 120 minutes, or two hours. After 30 minutes, the competitor’s rate of CBD absorption could barely be detected, which is a striking contrast from the aforementioned longevity of the TurboCBD™ dose.
Lexaria Bioscience has already licensed the use of DehydraTECH to multiple companies, though it has had 16 patents already granted for the innovative work they’ve done in the drug delivery platform sector. There are still over 60 patents pending. Consumers can learn more about Lexaria by visiting www.lexariabioscience.com.






