Medical Research
InMed Finds Cannabinol (CBN) Cannabinoid May Benefit a Rare Skin Condition and Glaucoma
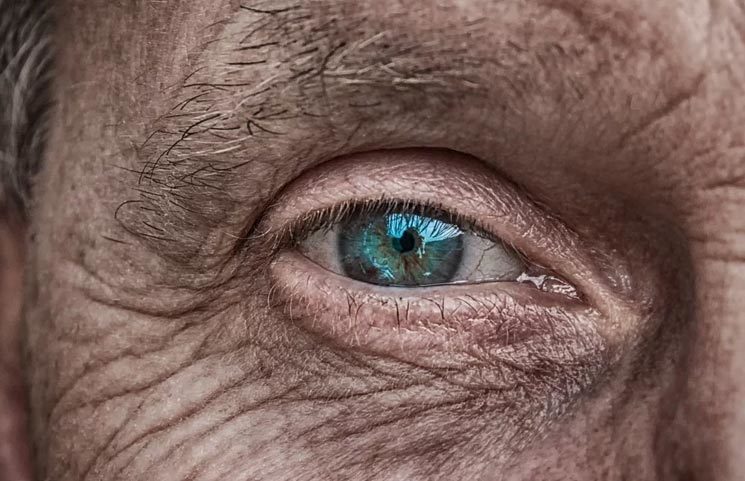
A Biopharmaceutical company specializing in cannabinoid-based therapeutics, InMed Pharmaceuticals found that a lower concentrated cannabinol (CBN) within their formulas were effective in treatments involving skin conditions and eye-related disease reports Mugglehead.
InMed first got the approval to commence clinical trials of INM-755 in December 2019 after the regulatory and ethics committee gave their approval. Today, the INM-755 formula is deemed effective in treating epidermolysis bullosa, which is a rare type of skin condition. Similarly, the INM-088 formula garnered their attention, as it was effective in treating glaucoma.
NEWS: InMed First to Advance #Cannabinol #CBN into Therapeutic Clinical Trials#rarecannabinoids #cannabinoids #therapeuticcannabinoidshttps://t.co/kiIqvm4IbZ
— InMed Pharmaceuticals (@inmedpharma) January 20, 2020
Speaking in regard to the achievement attained is the CEO of InMed, Eric Adams who shared that over 30 preclinical pharmacology and toxicology studies have been conducted by the firm, who’ve reasoned that CBN’s therapeutic benefits shouldn’t be taken so lightly. In particular, he said:
“We are proud to be the first company to investigate cannabinol’s therapeutic potential in human clinical trials […] Identifying potential therapeutic advantages of CBN in specific disease models over […] THC and CBD.”
He also disclosed that moving forward, more biosynthetic manufacturing methods would be considered and therapeutic uses of CBN would be analyzed to address other health conditions.






